BETA IMPACTS OUTCOMES
FUNCTIONAL BETA CELLS CAN IMPACT CLINICAL OUTCOMES1-6
ROLE IN GLYCEMIC CONTROL
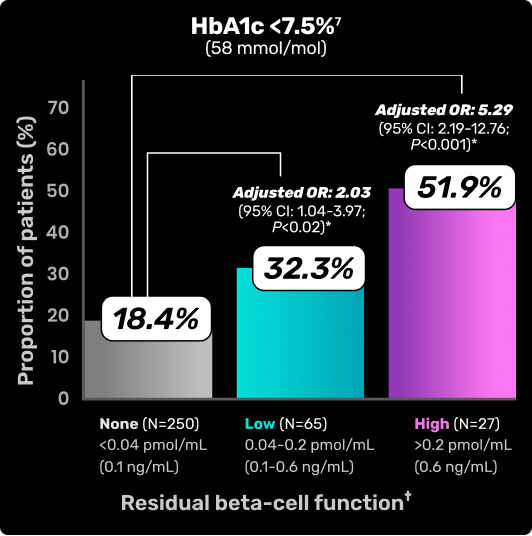
RESIDUAL BETA-CELL FUNCTION IS ASSOCIATED WITH LOWER HBA1C LEVELS8
Chart depicts HbA1C levels based on residual beta-cell function as measured by C-peptide.8
Study overview and limitations: Results from Danish Study Group for Childhood Diabetes (DanDiabKids) evaluating 342 pediatric patients aged 4.8-18.9 years with T1D for 3 to 6 years. Limitations were reported for this study. Thirty-nine percent of children eligible for the study according to the DanDiabKids registry did not participate. The HbA1c used for the study was analyzed locally.8
*Logistic regression analysis adjusted for sex, age, pubertal status, diabetes duration, and insulin administration technique.8
†Standard meal–stimulated C-peptide was used to assess residual beta cell function.8
Figure adapted from Sørensen JS, et al. Diabetes Care. 2013.
CI=confidence interval; HbA1c=hemoglobin A1c; OR=odds ratio.
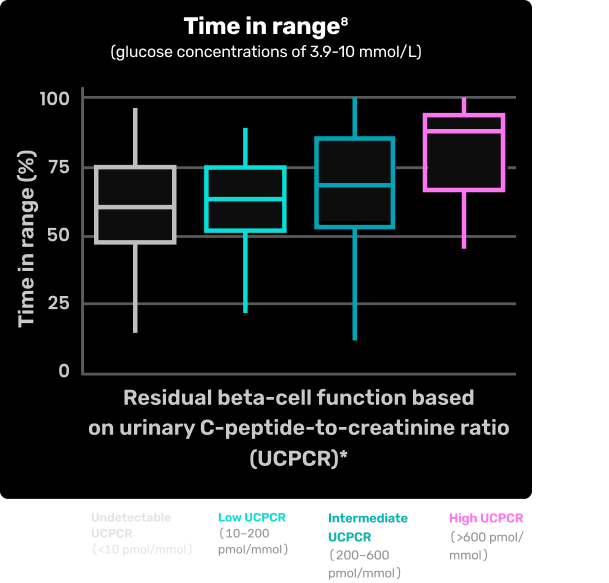
ASSOCIATION OF BETA-CELL FUNCTION AND TIME IN RANGE8
Chart depicts time in range based on residual beta-cell function as measured by urinary C-peptide-to-creatinine ratio (UCPCR)8
Study overview and limitations: Results of a cross-sectional cohort study examining the association between residual beta-cell function and glucose control (as metrics of continuous glucose monitoring) in patients with T1D (N=499). Limitations were reported for this study. First, because of the cross-sectional design, it is impossible to make claims of causality. Second, the UCPCR is not considered as gold standard for measuring beta-cell function. Therefore, this study may underestimate the contribution of beta-cell function to CGM-derived metrics and HbA1c. Finally, in the Netherlands, individuals with inadequate glycemic control generally have access to more devices. It is seen that participants with less beta-cell function more often use pumps, but did not obtain a better TIR, suggesting that the association between TIR and UCPCR may be further underestimated in this cohort of individuals with access to advanced tools of diabetes management.8
*Estimated using post-meal urinary C-peptide–to–creatinine ratio (UCPCR): undetectable (<0.01 nmol/mmol), low (0.01-0.2 nmol/mmol), intermediate (0.2-0.6 nmol/mmol), or high (>0.6 nmol/mmol).8
Figure adapted from Fuhri Snethlage CM, et al. Diabetes Care. 2024
BMI=body mass index; CGM=continuous glucose monitoring; HbA1c=hemoglobin A1c; T1D=type 1 diabetes; TIR=time in range.
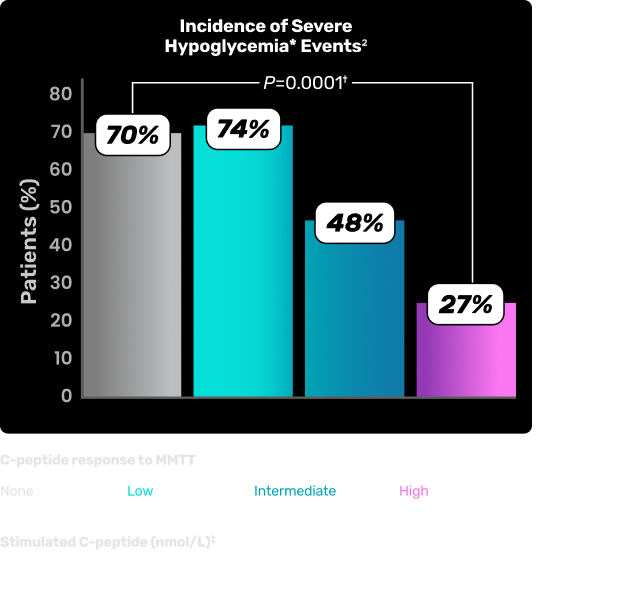
ASSOCIATION OF RESIDUAL BETA-CELL FUNCTION AND INCIDENCE OF SEVERE HYPOGLYCEMIC EVENTS1-6
Chart depicts incidence of severe hypoglycemic events with a mean of 6.5 years of follow-up based on residual beta-cell function as measured by C-peptide levels.2¶
Figure adapted from Gubitosi-Klug RA, et al. J Clin Invest. 2021.
Study overview and limitations: An analysis conducted within the DCCT- EDIC cohort that evaluated residual beta-cell function in individuals with a mean T1D duration of 35 years. Beta-cell function was assessed through C-peptide concentrations, measured at 9 time points during a 4-hour MMTT. Participants were categorized by peak C-peptide concentration (nonresponder, low, intermediate, or high), and associations with metabolic outcomes, severe hypoglycemia, and microvascular complications were examined. No limitations were reported for this study.2,5
Data from the DCCT/EDIC studies, which included study participants from US and Canada. C-peptide studies, were carried out between 2015 and 2017.
*Hypoglycemia requiring assistance.2
†Based on Cochran–Armitage trend test.2
‡1 nmol/L C-peptide=3.0203 ng/mL.
§During a 4-hour MMTT, all C-peptide values were <0.003 nmol/L, which was the lower limit of detection for the C-peptide assay used in the study.2
||Peak C-peptide during 4-hour MMTT.2
¶During the DCCT study, participants were asked to report all episodes of suspected severe hypoglycemia immediately, and all participants were interviewed regarding the episodes using a standard series of questions. During the EDIC study, the ascertainment of events was restricted to the 3-month window prior to the annual visit and used the same standardized questions.2
DCCT=Diabetes Control and Complications Trial; EDIC=Epidemiology of Diabetes
Interventions and Complications; MMTT=mixed-meal tolerance test; T1D=type 1 diabetes.
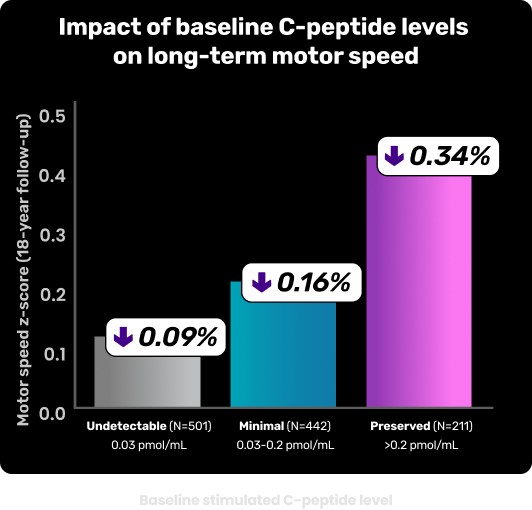
ASSOCIATION OF RESIDUAL BETA-CELL FUNCTION AND MOTOR SPEED PRESERVATION11
After an 18-year follow-up, motor speed z-score increased with higher baseline stimulated C-peptide levels11
Data presentation adapted from Wang T, et al. Diabetes. 2025.
Study overview: An analysis conducted of 1144 participants within the DCCT/EDIC aimed to determine whether baseline-stimulated C-peptide levels are associated with cognitive performance over an 18-year follow-up in adults with T1D. Baseline-stimulated C-peptide levels were measured using standardized assays and cognitive performance was assessed using a comprehensive neuropsychological test battery. Multivariable linear regression models were used to evaluate the association between baseline-stimulated C-peptide and cognitive z-scores adjusting for covariates including demographics, HbA1c, and diabetes-related complications (retinopathy, neuropathy, nephropathy).11*
*During the DCCT study, participants were asked to report all episodes of suspected severe hypoglycemia immediately, and all participants were interviewed regarding the episodes using a standard series of questions. During the EDIC study, the ascertainment of events was restricted to the 3-month window prior to the annual visit and used the same standardized questions.2
DCCT=Diabetes Control and Complications Trial; EDIC=Epidemiology of Diabetes Interventions and Complications; HbA1c=hemoglobin A1c; T1D=type 1 diabetes.
IMPACT ON OUTCOMES
Reduced risk of retinopathy and nephropathy are associated with residual beta-cell function.9
risk reduction of retinopathy observed
in patients with higher C-peptide levels >0.6 ng/mL9
(OR 0.55 [95% CI 0.34-0.89], P=0.014)
risk reduction of nephropathy observed
in patients with C-peptide levels >0.6 ng/mL9
(OR 0.61 [95% CI 0.38-0.96], P=0.033)
Data presentation adapted from Harsunen M, et al. Lancet Diabetes Endocrinol. 2023.
Study overview and limitations: Results of a Finnish study combining longitudinal follow-up data from 847 newly diagnosed individuals with T1D and cross-sectional long-duration data in 3984 participants from FinnDiane and 645 participants from DIREVA to investigate residual insulin secretion in patients with T1D and evaluate C-peptide levels over time and their link to complications (N=5586). This study has several limitations, including gaps in data due to its retrospective, longitudinal design and small sample sizes in the longitudinal cohorts. The C-peptide assay changed over time, and measurements were taken under nonstandardized conditions. The analysis may be subject to unmeasured and residual confounding, as well as potential selection bias in hazard ratio estimates.9
Data from the DCCT/EDIC studies.
DCCT=Diabetes Control and Complications Trial; EDIC= Epidemiology of Diabetes Interventions and Complications; T1D= type 1 diabetes.
LONG-TERM DATA ARE AVAILABLE TO SHOW THE ASSOCIATION OF C-PEPTIDE LEVELS WITH OUTCOMES IN T1D12
In the SDRNT1BIO study, a follow-up of ~11 years showed C-peptide levels and association with12:
Incidence of DKA
Hypoglycemia requiring
hospitalization
Retinopathy and maculopathy
Study overview and limitations: Long-term observational study of 5630 adults with autoimmune T1D from the Scottish Diabetes Research Network Type 1 Bioresource (SDRNT1BIO) cohort, assessing associations between baseline C-peptide levels and incident clinical outcomes over 11 years of follow-up using electronic health records and adjusted statistical models. Higher C-peptide at baseline was associated with a reduced risk of DKA, hypoglycemia requiring hospitalization, and transition to retinopathy or maculopathy. Long-term observational studies do not have control of variables, are vulnerable to bias, and cannot prove causation.12
DKA=diabetic ketoacidosis; T1D=type 1 diabetes.
EXPLORING THE BENEFITS AND FEASIBILITY OF...
A range of investigational mechanisms are currently being studied including, but not limited to:
Downregulation and disrution of TID autoimmune mechanisms13
Improving insulin sensitivity14
Regeneration and replacement of functional beta cells15
References
-
Jeyam A, Colhoun H, McGurnaghan S, et al; SDRNT1BIO Investigators. Clinical impact of residual C-peptide secretion in type 1 diabetes on glycemia and microvascular complications. Diabetes Care. 2021;44(2):390-398.
-
Gubitosi-Klug RA, Braffett BH, Hitt S, et al; DCCT/EDIC Research Group. Residual β cell function in long-term type 1 diabetes associates with reduced incidence of hypoglycemia. J Clin Invest. 2021;131(3):e143011.
-
Palmer JP, Fleming GA, Greenbaum CJ, et al. C-peptide is the appropriate outcome measure for type 1 diabetes clinical trials to preserve β-cell function: report of an ADA workshop, 21-22 October 2001. Diabetes. 2004;53(1):250-264.
-
Lachin JM, McGee P, Palmer JP; DCCT/EDIC Research Group. Impact of C-peptide preservation on metabolic and clinical outcomes in the Diabetes Control and Complications Trial. Diabetes. 2014;63(2):739-748.
-
Steffes MW, Sibley S, Jackson M, et al. β-cell function and the development of diabetes-related complications in the diabetes control and complications trial. Diabetes Care. 2003;26(3):832-836.
-
Nathan DM. Realising the long-term promise of insulin therapy: the DCCT/EDIC study. Diabetologia. 2021;64(5):1049-1058.
-
Fuhri Snethlage CM, McDonald TJ, Oram RD, et al. Residual β-cell function is associated with longer time in range in individuals with type 1 diabetes. Diabetes Care. 2024;47(7):1114-1121.
-
Sørensen JS, Johannesen J, Pociot F, et al; Danish Society for Diabetes in Childhood and Adolescence. Residual β-cell function 3-6 years after onset of type 1 diabetes reduces risk of severe hypoglycemia in children and adolescents. Diabetes Care. 2013;36(11):3454-3459.
-
Harsunen M, Haukka J, Harjutsalo V, et al. Residual insulin secretion in individuals with type 1 diabetes in Finland: longitudinal and cross-sectional analyses. Lancet Diabetes Endocrinol. 2023;11(7):465-473.
-
Harsunen M, Haukka J, Harjutsalo V, et al. Residual insulin secretion in individuals with type 1 diabetes in Finland: longitudinal and cross-sectional analyses. Lancet Diabetes Endocrinol. 2023;11(7)(suppl):1-13
-
Leighton E, Sainsbury CAR, Jones GC. A practical review of C-peptide testing in diabetes. Diabetes Ther. 2017;8(3):475-487.
-
Mellor JH, Blackburn LAK, McGurnaghan SJ, et al. Long-term effect of C-peptide on clinical outcomes in the Scottish Type 1 Bioresource cohort. Abstract presented at: IDS 2024 Congress; November 7-8, 2024; Bruges, BE. Accessed March 7, 2026. https://idsbruges2024.com/wp-content/uploads/2024/10/IDS-BOA_PDF-2.pdf
-
Von Scholten BJ, Kreiner FF, Gough SCL, et al. Current and future therapies for type 1 diabetes. Diabetologia. 2021;64(5):1037-1048.
-
Nowak C, Lind M, Sumnik Z, et al. Intralymphatic GAD-Alum (Diamyd®) improves glycemic control in type 1 diabetes with HLA DR3-DQ2. J Clin Endocrinol Metab. 2022;107(9):2644-2651.
-
Pociot F. Capturing residual beta cell function in type 1 diabetes. Diabetologia. 2019;62(1):28-32.
MAT-US-2504445-v2.0-03/2026