AUTOIMMUNE MECHANISM OF T1D
T1D IS MORE THAN LACK OF INSULIN-IT’S DEFINED BY THE IMPACT OF BETA-CELL DESCRUCTION
T1D is an autoimmune condition characterized by the progressive and irreversible loss of beta cells1,2
MECHANISM OF DISEASE
Autoimmunity in action: T cell-driven pathogenesis of autoimmune T1D3-8
An autoimmune trigger initiates loss of immune tolerance.*
Antigen-presenting cells (APCs) process beta-cell autoantigens and migrate to lymph nodes.
In the lymph nodes, APCs drive clonal expansion of :
Autoreactive CD4+
T cells (helper T cells)
CD4+ T cells4-6:
-
promote responses by effector CD8+ T cells
-
stimulate autoantibody production
Autoreactive CD8+
T cells (cytotoxic T cells)
CD8+ T cells4-6:
-
mediate beta-cell death in a contact-dependent manner through perforin and granzyme secretion
B cells
B cells4-6:
-
act as APCs to activate autoreactive T cells
*The precise mechanism that leads to the loss of immune tolerance has not been fully defined.6
T1D=type 1 diabetes.
IMPACT OF AGE ON T1D PROGRESSION
The younger a person is at the onset of T1D, the greater the severity of beta-cell destruction9-12
THE AGGRESSIVENESS OF INSULITIS TYPICALLY VARIES WITH AGE...
Patients receiving a diagnosis before the age of 7 years display a more aggressive profile than patients aged 13 years or older.9,11
...RESULTS IN DIFFERENTIAL RATES OF BETA LOSS
In young patients, beta-cell loss is rapid and extensive; those diagnosed in their teens or later may still retain a measurable beta-cell reserve.9-11
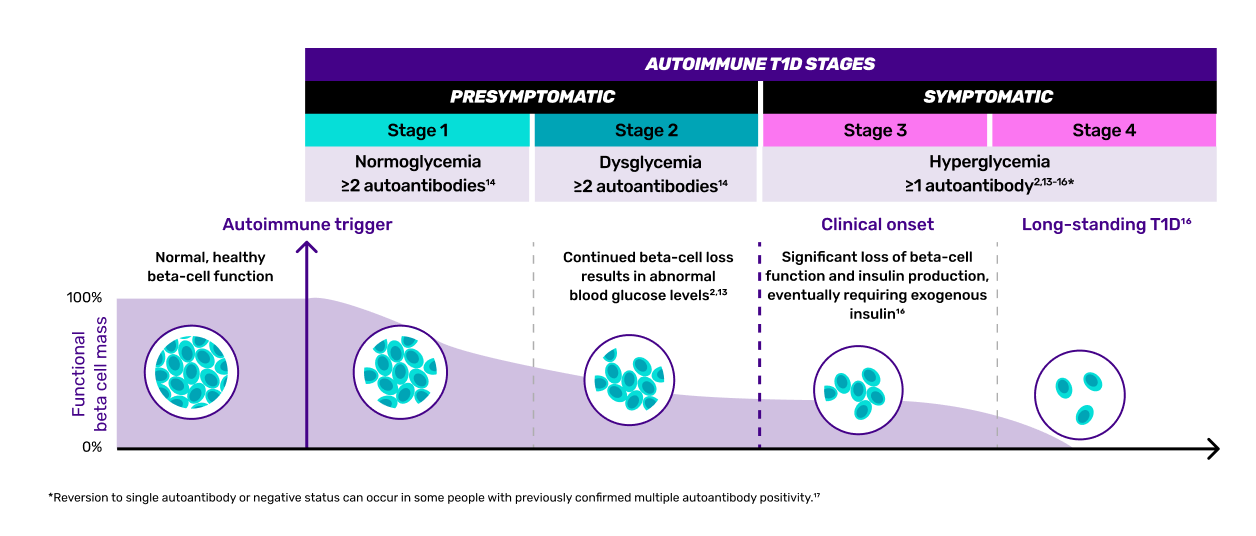
BETA-CELL DESTRUCTION IS A WARNING SIGN OF LOSS OF INSULIN PRODUCTION, EVEN IF THE PATIENT SHOWS NO SYMPTOMS1,2,13
Many patients still have some beta-cell function in Stage 3 T1D: beta-cell function decline presents differently and happens at different rates1,2
THE HONEYMOON PHASE IN STAGE 313:
-
A temporary period of improved blood sugar control that occurs in some people with newly diagnosed Stage 3 T1D who are just starting insulin
-
The pancreas may still produce some insulin, which may reduce the amount of external insulin injections needed
Duration and characteristics:
-
Typically lasts for a few weeks to a few years
-
Blood sugar levels may be easier to control, requiring less insulin
-
Endogenous insulin may reduce the amount of external insulin needed, but only for a short period of time
-
The duration and intensity of the honeymoon phase vary widely among individuals
EVEN REMAINING BETA CELLS CAN HAVE AN IMPACT ON GLYCEMIC REGULATION

Residual beta-cell function can reduce risk of T1D complications, including21-28:
-
Hypoglycemia
-
Retinopathy
-
Nephropathy
-
Neuropathy

Residual beta cells can help maintain glucose homeostasis to impact the likelihood of time in range and achieving target hemoglobin A1c (HbA1c)21,29

Research on T1D suggests that timely disease management approaches may influence cellular processes, which could potentially impact disease progression and long-term outcomes28,30,31
NOT MEETING GLYCEMIC TARGETS IS ASSOCIATED WITH DIABETES-RELATED STRESS34
Key factors34:

Suboptimal glycemic control

Potential long-term complications

Fear of hypoglycemia

Self-care burden
References
-
Scheiner G, Weiner S, Kruger D, Pettus J. Screening for type 1 diabetes: role of the diabetes care and education specialist. ADCES Pract. 2022;10(5):20-25.
-
Insel RA, Dunne JL, Atkinson MA, et al. Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care. 2015;38(10):1964-1974.
-
Addissouky TA, Ali MMA, El Sayed IET, et al. Type 1 diabetes mellitus: retrospect and prospect. Bull Natl Res Cent. 2024;48(42):1-15.
-
Burrack AL, Martinov T, Fife BT. T cell-mediated beta cell destruction: autoimmunity and alloimmunity in the context of type 1 diabetes. Front Endocrinol. 2017;8:343.
-
Houeiss P, Luce S, Boitard C. Environmental triggering of type 1 diabetes autoimmunity. Front Endocrinol. 2022;13:933965.
-
Nagy G, Szekely TE, Somogyi A, et al. New therapeutic approaches for type 1 diabetes: disease-modifying therapies. World J Diabetes. 2022;13(10):835-850.
-
Sauls RS, McCausland C, Taylor BN. Histology, T-cell lymphocyte. In: StatPearls. StatPearls Publishing; 2023.
-
Pugliese A. Insulitis in the pathogenesis of type 1 diabetes. Pediatr Diabetes. 2016;17(suppl 22):31-36.
-
Leete P, Mallone R, Richardson SJ, et al. The effect of age on the progression and severity of type 1 diabetes: potential effects on disease mechanisms. Curr Diab Rep. 2018;18(11):115.
-
Marcovecchio ML, Hendriks AEJ, Delfin C, et al; INNODIA consortium. The INNODIA Type 1 Diabetes Natural History Study: a European cohort of newly diagnosed children, adolescents and adults. Diabetologia. 2024;67(6):995-1008.
-
Leete P, Willcox A, Krogvold L, et al. Differential insulitic profiles determine the extent of β-cell destruction and the age at onset of type 1 diabetes. Diabetes. 2016;65(5):1362-1369.
-
Poudel A, Savari O, Striegel DA, et al. Beta-cell destruction and preservation in childhood and adult onset type 1 diabetes. Endocrine. 2015;49(3):693-702.
-
Understanding A1C diagnosis. American Diabetes Association. Accessed March 6, 2026. https://www.diabetes.org/a1c
-
Phillip M, Achenbach P, Addala A, et al. Consensus guidance for monitoring individuals with islet autoantibody-positive pre-stage 3 type 1 diabetes. Diabetes Care. 2024;47(8):1276-1298.
-
American Diabetes Association Professional Practice Committee. 2. Diagnosis and classification of diabetes: standards of care in diabetes—2025. Diabetes Care. 2025;48(suppl 1):S27-S49.
-
The stages of type 1 diabetes and why they’re important. Breakthrough T1D. Accessed March 7, 2026. https://breakthrought1d.org.au/app/uploads/2025/03/Stages-of-T1D-V2.pdf
-
American Diabetes Association Professional Practice Committee for Diabetes. Standards of care in diabetes—2026. Diabetes Care. 2026;49(suppl 1):S1-S371.
-
Gitelman SE, Evans-Molina C, Guolo A, et al. β-cell glucose sensitivity to assess changes in β-cell function in recent-onset stage 3 type 1 diabetes. Diabetes. 2023;72(9):1289-1296.
-
Kawasaki E. Anti-islet autoantibodies in type 1 diabetes. Int J Mol Sci. 2023;24(12):10012.
-
Wang YN, Li R, Huang Y, et al. The role of B cells in the pathogenesis of type 1 diabetes. Front Immunol. 2024;15:1450366.
-
Sørensen JS, Johannesen J, Pociot F, et al; Danish Society for Diabetes in Childhood and Adolescence. Residual β-cell function 3-6 years after onset of type 1 diabetes reduces risk of severe hypoglycemia in children and adolescents. Diabetes Care. 2013;36(11):3454-3459.
-
Gubitosi-Klug RA, Braffett BH, Hitt S, et al; DCCT/EDIC Research Group. Residual β cell function in long-term type 1 diabetes associates with reduced incidence of hypoglycemia. J Clin Invest. 2021;131(3):e143011.
-
Jeyam A, Colhoun H, McGurnaghan S, et al; SDRNT1BIO Investigators. Clinical impact of residual C-peptide secretion in type 1 diabetes on glycemia and microvascular complications. Diabetes Care. 2021;44(2):390-398.
-
Palmer JP, Fleming GA, Greenbaum CJ, et al. C-peptide is the appropriate outcome measure for type 1 diabetes clinical trials to preserve β-cell function: report of an ADA workshop, 21-22 October 2001. Diabetes. 2004;53(1):250-264.
-
Lachin JM, McGee P, Palmer JP; DCCT/EDIC Research Group. Impact of C-peptide preservation on metabolic and clinical outcomes in the Diabetes Control and Complications Trial. Diabetes. 2014;63(2):739-748.
-
Steffes MW, Sibley S, Jackson M, et al. β-cell function and the development of diabetes-related complications in the diabetes control and complications trial. Diabetes Care. 2003;26(3):832-836.
-
Leighton E, Sainsbury CAR, Jones GC. A practical review of C-peptide testing in diabetes. Diabetes Ther. 2017;8(3):475-487.
-
Nathan DM. Realising the long-term promise of insulin therapy: the DCCT/EDIC study. Diabetologia. 2021;64(5):1049-1058.
-
Fuhri Snethlage CM, McDonald TJ, Oram RD, et al. Residual β-cell function is associated with longer time in range in individuals with type 1 diabetes. Diabetes Care. 2024;47(7):1114-1121.
-
Islam K, Islam R, Nguyen I, et al. Diabetes mellitus and associated vascular disease: pathogenesis, complications, and evolving treatments. Adv Ther. 2025;42(6):2659-2678.
-
Noso S, Babaya N, Hiromine Y, et al. Metabolic signatures of β-cell destruction in type 1 diabetes. J Diabetes Investig. 2023;14(1):48-57.
-
Kramer CK, Retnakaran R, Zinman B. Insulin and insulin analogs as antidiabetic therapy: a perspective from clinical trials. Cell Metab. 2021;33(4):740-747.
-
Laffel LM, Kanapka LG, Beck RW, et al; CGM Intervention in Teens and Young Adults with T1D (CITY) Study Group. Effect of continuous glucose monitoring on glycemic control in adolescents and young adults with type 1 diabetes: a randomized clinical trial. JAMA. 2020;323(23):2388-2396.
-
Rodriguez-Munoz A, Picon-Cesar MJ, Tinahones FJ, et al. Type 1 diabetes–related distress: current implications in care. Eur J Intern Med. 2024;125:19-27.
MAT-US-2504444-v2.0-03/2026